Dr. ADIL K. SALLOM, MRCS, D.MAS
Member Royal College ofSsurgeons of Ireland Fellow ship of Arab board of medical specialization Member of world association of laparoscopic surgeons Diploma in general surgery - Iraq Diploma in Minimal Access surgery
Project Submitted Towards Completion Of Diploma In Minimal Access Surgery, World Laparoscopy Hospital, Gurugram, NCR Delhi, India. AUGUST 2007.
Abstract:
The most common surgical procedure performed for treatment of GERD is a Nissen fundoplication. Fundoplication means fundus of the stomach which is on left of the oesophagus and main portion of the stomach is wrapped around back of the oesophagus. Recognizing the relationship between a dysfunctional lower esophageal sphincter and GERD, Dr. Nissen developed this procedure in the early twentieth century. However, it carried with it, a significant morbidity rate. With the success of laparoscopy used to remove the gallbladder in the late 1980's, surgeons began applying this technology to other surgical diseases. With some modifications, the Nissen fundoplication is now performed using minimally invasive techniques. Patients have small incisions, have less pain, leave the hospital sooner, and return to normal activity sooner. Furthermore, surveys following laparoscopic Nissen fundoplication have demonstrated immense patient satisfaction with the procedure
Key words : Esophagus, anti-reflux surgery, laparoscopy, GERD
Introduction :
GASTROESOPHAGEAL reflux disease is a very common condition for which most patients are treated with medical treatment like proton pump inhibitors and prokinetic agents. However, 20% of patients who develop complications or experience recurrent symptoms after discontinuing medical therapy require surgical treatment. Conventional surgical management of gastroesophageal reflux disease with fundoplication is effective, durable, and in some aspects superior to medical therapy. The ability to perform this procedure laparoscopically may further the applications of this treatment. Laparoscopic fundoplication has been shown to be safe and as effective as open fundoplication, and has become the preferred surgical option.
Objective
To compare the results of open and laparoscopic fundoplication: The aims of this study is evaluating the efficacy, safety and outcome of laparoscopic surgery for GERD in comparison with open surgery, and also determine the risk factors which influence the outcome to identify those patients that are not good candidates for laparoscopic approach. The following parameters were evaluated for laparoscopic and open procedure: 1-Operating technique 2-Operating time 3-Intraoperative complications 4- Risk of anesthesia 5-Rate of conversion to open surgery 6-Postoperative pain and the opiate analgesic requirements 7-Postoperative morbidity & mortality 8-Hospital stay 9-Cost 10-Quality of life analysis Materials and Methods : A literature search was performed using search engine Google, HighWire Press, SpringerLink and library facility available at Laparoscopic Hospital. The following search terms was used: laparoscopic fundoplication versus open surgery . Criteria for selection of papers were upon statistical way of analysis, institute if they were specialized for laparoscopy, the way of management and the technique of opertions. GERD : Gastroesophageal reflux disease (GERD) is one of the most common gastrointestinal problems in the western countries. GERD is resulting from the movement of gastric contents into the esophagus. It has been reported that more than 42% of Americans experience symptoms of GERD at least once a month, and up to 10% of the population experience daily symptoms (1-4).
Though exact figures are unknown, it is estimated that 16 - 40 million adults in the United States suffer from GERD (5). The prevalence of GERD has increased dramatically this century, as esophagitis was at one time a very rare finding at autopsy (6). The increasing prevalence of reflux is probably related to changes in diet and lifestyle .
Symptoms : The most common symptom of GERD is heartburn. Heartburn (pyrosis),which is experienced as a retrosternal burning discomfort,most commonly after meals and when supine. This is due to acid stimulation of sensory nerve endings in the deeper layers of the esophageal epithelium.
Another most common symptom is regurgitation. Regurgitation is the return of gastric or esophageal fluid into the pharynx without nausea, retching, or abdominal contractions. Regurgitation often occurs at night while the patient is sleeping. When this results in a sudden awakening with hot fluid in the throat. It is often associated with coughing, choking, and shortness of breath due to aspiration of the regurgitated fluid. It can also occur after a large meal.
A third symptom is dysphagia, which is occurs in about one third of people with GERD. It can be caused by a peptic stricture due to oesophagitis. Atypical symptoms . These include chest pain, wheezing, hoarseness, chronic cough, choking, globus, and dental caries. These symptoms are caused by injury of the esophagus, larynx, airways, pharynx, or teeth due to refluxed fluid from the stomach.
Diagnosis :
Clinical history
24-pH study
Endoscopy
Manometry
Pathophysiology :
The cause of GERD is multifactorial. GERD can be caused by a failure of intrinsic mechanisms of: the lower esophageal sphincter (LES), the function of the esophageal body, and the function of the gastric reservoir. Though the esophagus must be able to clear acid normally, and the stomach should be able to empty normally, a weak LES is the most important contributor to GERD.
To function appropriately, the LES must have a normal length, normal pressure, normal relaxation, and be located intra-abdominal below the diaphragmatic crura. There, the intra-abdominal pressure can assist the action of the LES. The LES pressure is commonly low in patients with GERD but sometime may be normal or even elevated. The most important factor causing GERD i s transieint relaxation of the LES, by far is the most important cause of reflux . Hiatus hernia type I one may give rise to GERD. A hiatus hernia type I is a common abnormality in which the esophagogastric junction and some part of the fundus of stomach lie above the diaphragm and in the chest. It has been estimated that hiatus hernia occurs in more than 10% of the general population, but not every patient with hiatus hernia must have GERD and the reverse is also true. It should be noted that up to 80% of patients with symptomatic reflux have a hiatus hernia, and patients with erosive esophagitis mostly associated with hiatus hernia.
Gastric acid reflux is an essential component of GERD, but the reflux is a combination of gastric contents and duodenal contents in 50% (7). Acid-only reflux occurs in only 40%. Gastric juice contains both HCl and the enzyme pepsin, while duodenal juice contains alkaline bile acid, and pancreatic juice. Acid, pepsin, and bile salts all interact together and contribute to erosive esophagitis. It has been found that acid and bile, causing more severe mucosal damage to the esophagus than acid alone.
Complications: Complications develop in about 20% of patients with GERD (8). Complications of GERD include the esophageal complications and the effects on the respiratory tract (aspiration pneumonia, asthma, pulmonary fibrosis). Esophageal effect include erosive esophagitis, esophageal ulcers, stricture, and Barrettís esophagus which occur in 10%.
Barrettís esophagus is the metaplastic change of normal squamous epithelium to columnar epithelium, it is a premalignant condition. There is evidence that Barrett¹s esophageal mucosa is prone to develop adenocarcinoma of the esophagus. Barrett¹s mucosa places the patient at increased risk (30 fold to 350 fold) for the subsequent development of esophageal adenocarcinoma (9,10). In patients with Barrett¹s mucosa, the risk of developing carcinoma is about 1% per year. Previously, adenocarcinoma of the esophagus accounted for fewer than 8% of all esophageal tumors, but it now accounts for at least 50% of esophageal cancers. It has been suggested that this rising in incidence may be due to the increasing occurrence of Barrett¹s metaplasia. The increased prevalence of adenocarcinoma at the gastroesophageal junction is a matter of concern, and seems to be related to inadequate control of gastroesophageal reflux (10,11).
Treatment (non surgical) :
Non surgical treatment includes lifestyle modifications and medications (12-14). Lifestyle modifications include elevation of the head of the bed during sleeping and avoidance of eating for 2 to 3 hours before sleeping. Avoidance of fatty or spicy food, cessation of smoking,caffeine intake and alcohol , may significantly improve symptoms. However, lifestyle and dietary changes are successful in only 20% of patients.
Most symptomatic patients treat themselves with over the counter medications, such as antacids or H2-receptor antagonists. Only a small percentage of people who actually experience GERD consult a physician. H2-receptor antagonist use in standard doses can achieve symptomatic relief in 25% to 60% of patients, and endoscopic resolution of esophagitis in 50%. Use of high dose H2-receptor antagonists can result in healing rates of 45-75%. Proton pump inhibitors (PPI) are the most effective medical therapy to heal esophagitis and control symptoms. The standard doses of PPIis resolves symptoms in 80-90% of patients, and heals the esophagitis in upto 90%. Larger doses are mainly required in patients with high-grade esophagitis. However, GERD is a chronic condition and disease tend to relapse if the drug dose is stopped, decreased, or sometimes even if a dose is skipped. Esophagitis relapse up to 80% of the time within six months, both symptomatically and by endoscopy, if PPI therapy is stopped or the drug dose is decreased.
Surgical Treatment : The most common cause of GERD is a defective LES. So the medication is not always so effective, specially when LOS is defective or in patients present with a hiatus hernia. ,Also this therapy will do nothing to prevent the effect of pepsin and duodenal contents on the esophageal mucosa. Surgery is the treatment of choice that can prevent esophageal exposure to both gastric and duodenal juices. Antireflux surgery also repairs the hiatal hernia, WHICH a significant contributor to the reflux. Indications for surgical therapy: There is general agreement in both the medical and surgical literature on the indications for surgical therapy in the treatment of GERD (12,13,16-19), so surgical therapy is indicated in patient who:
- Refractory to medical treatment which occurs in about 10% of patients. These patients have persistent, symptomatic esophagitis that are resistant to medical treatment.
- Present with complication of GERD that occurs in 20% of patients. These include continued esophagitis, grade 3 or 4 esophagitis, esophageal ulcers, esophageal stricture, and Barrettís esophagus.
- In tolerance to medical treatment.
- Atypical symptoms which are primarily pulmonary and laryngeal. These include asthma, chronic cough, hoarseness, laryngitis, chest pain, and recurrent aspiration. Although the outcome of surgery in patients with extraesophageal symptoms is, in general, less successful than in patients with typical symptoms, patients tend to require less corticosteroid treatment for asthma after surgery. The best surgical results are in those patients with a good response to PPIis.
- A symptomatic hiatal hernia or paraesophageal hernia deserve consideration for surgical repair to correct the hernia. The indications for antireflux surgery have not really changed, but patient and physician acceptance of a minimal access surgical procedure has increased.
Evaluation of patient prior to surgery : The major goal of the preoperative evaluation is to make a definitive diagnosis. EGD with biopsy and/or 24 hr. pH studies are the only tests that can objectively prove reflux. Tests to assess the motor function of the esophagus, and perhaps the stomach, are also needed .
EGD : EGD is performed to look for esophagitis and complications of GERD such as stricture or Barrettís metaplasia. EGD may also detect hiatal hernia. 24 hr pH monitoring is the most accurate test for the detection of GERD, but is only 85-90% accurate. It should be performed liberally, but is probably not needed in those patients with typical symptoms of GERD and evidence of esophagitis on endoscopy. It is a useful test in patients with atypical symptoms or extraesophageal manifestations of GERD, or in patients with typical symptoms of GERD but who have normal findings on endoscopy. Unfortunately, patients should stop taking PPIs for 10-14 days prior to testing. Esophageal Manometry : Esophageal manometry is performed to evaluate the strength and adequacy of both the upper and lower esophageal sphincters, as well as the strength and coordination of peristalsis in the body of the esophagus. It is the definitive test for primary motor disorders of the esophagus, such as achalasia , nutcracker esophagus,and sclerosis. In patients with GERD, esophageal manometry determines that 10-15% of patients have poor esophageal motility and may require a partial (Toupet) fundoplication (see below). Patients with GERD tend to have low LES pressures, but they may also be normal or high .Barium Esophagram: A barium esophagram is the test of choice to evaluate dysphagia ,hiatal hernia or paraesophageal henia. Barium esophagram is also sensistive test for detection of esophageal strictures. It is not a good test for the dignosis of reflux. A gastric emptying study is important, if symptoms of gastric stasis such as nausea and emesis of retained food - are prominent, or if the patient is a diabetic, then a gastric emptying study will rule in or out delayed gastric emptying,a pyloroplasty or pylormyotomy may be hlepfull at the time of surgery if there is poor gastric emptying .
Antireflux surgery :
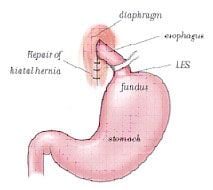
Antireflux surgery involves wrapping a portion of stomach (fundus) around LES with reduction of a hiatal hernia if present. This constructs a valve like mechanism to re-establish gastroesophageal junction competence. This creates a barrier to the reflux of gastric contents,so it provides relief of symptoms and prevents the complications associated with GERD.

Surgery for GERD was first performed in 1956, but because of the development of effective medical therapy, and the relatively smaller numbers of patients afflicted, surgical treatment was not widely performed between 1960 and 1990. In 1991, laparoscopic Nissen fundoplication was reported(16). Since that time, more than 10,000 laparoscopic antireflux procedures have been reported, and many more have been performed. The advantages of laparoscopic antireflux surgery are similar to those reported after other laparoscopic procedures, such as cholecystectomy. These include a short (one night) hospital stay, a earlier return to work and normal activities (two weeks), and fewer complications such as atelectasis, pneumonia, splenic injury, and fewer incisional hernias.
Laparoscopic antireflux surgery is technically challenging and should be performed only by surgeons with good training and with high experience in advanced laparoscopic surgical technique. There are two types of fundoplications that are most commonly performed: Nissen and Toupet Fundoplications. The Nissen operation involves wrapping the fundus completely around the esophagus (360¤), producing a short, loose wrap. In the Toupet operation, the fundus is wrapped only part of the way around the esophagus (270¤), producing a short, even looser wrap. The type of operation is chosen based on the severity of reflux and complications involved, as well as the function of the esophagus. While the Toupet results in less difficulty with gas bloat syndrome and swallowing, the Nissen procedure is the most effective for controlling reflux. Therefore, the Toupet is generally best for patients whose reflux is less severe . The latter is utilized in the small percent of patients who have severe dysmotility of the esophageal body. The mortality of laparoscopic procedure is essentially zero, and the morbidity is around 5%, which is less than after open surgery. The conversion rate to an open surgical technique is also less than 2%.
Most patients experience a mild degree of dysphagia postoperatively , which is slightly more in laparoscopic procedure than that of open operation, but this resolves in more than 95% of patients within the first month. The long-term incidence of postoperative dysphagia is 2% or less which is the same in both procedure . Other symptoms that the patient may experience early after the surgery include early satiety, hyperflatulence, bloating, and diarrhea. These symptoms also tend to be transient and resolve with time. The success of the operation in preventing reflux as determined by a 24 hr. pH study one year postoperatively is 93% (18). Patients with Barrettís esophagus prior to surgery still need surveillance endoscopy after surgery to evaluate for the development of dysplasia.
Techniques :
Fundoplication is done as either an open or a laparoscopic procedure. The open procedure involves an incision of about 20- 25 cm in the abdomen, while the laparoscopic approach is a minimally invasive technique producing 4 to 5( 0.5-1cm) incisions. Although the laparoscopic approach offers many advantages over the open technique, such as a quicker recovery and less complications, it may not be appropriate for some patients, including those who have had previous abdominal surgery or who have some pre-existing medical conditions. In addition, some patients may have to be converted from the laparoscopic procedure to the open technique during surgery. However, this is uncommon and most patients (95%) can undergo the laparoscopic procedure without difficulty.
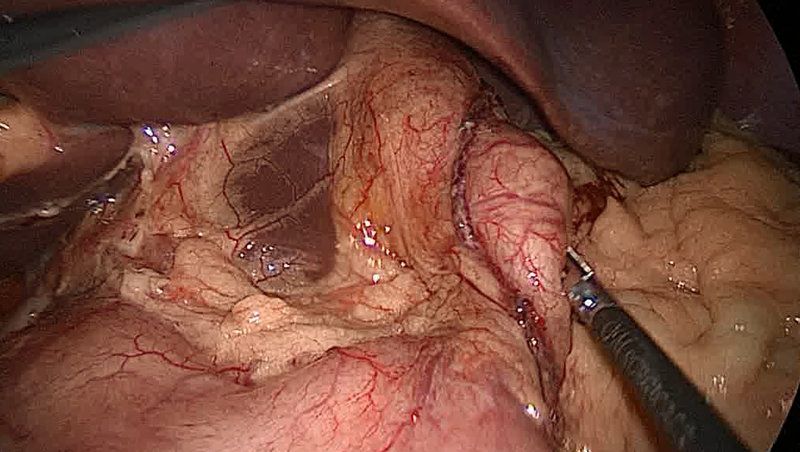
During the operation, the surgeon raises the liver to expose the junction between the stomach and the esophagus. A space is created behind the esophagus and the fundus of the stomach and freed from its attachment to the spleen(gastrosplenic ligament). The fundus is then pulled behind the esophagus and secured in place. Depending on the type of procedure, the wrap is either sutured to the esophagus itself (as in the Toupet), or it is sutured to the stomach on the other side of the wrap (as in the Nissen). If a hiatal hernia is present, it is repaired before.
In the laparoscopic procedure, surgeons use a trocar (a narrow tube-like instrument) to gain access to the abdomen. A laparoscope (a thin telescope connected to a video camera) is inserted through the trocar, giving the surgeon a magnified view of the patient's internal organs on a television monitor. Four additional trocars are then inserted to accommodate special instrumentation.So five trocars are used ,10 mm port camera is placed in midline 5cm above the umbilicus,5mm port is inserted in the right upper quadrant 8- 10 cm from the midline.The third 5- 10 mm port is placed in left upper quadrant -a mirror image of the right one. A further 5mm port is placed in the left anterior axillary line immediately below the costal margin .
Following the procedure, the incision(s) are closed with stitches or with surgical tape and bandages are applied.
Surgical Complications :
Perforation of the stomach or esophagus can occur in about 1% of patients. In rare cases, bleeding or perforation is not identified at the time of operation and a re-operation is necessary. Approximately 5% of patients require conversion to open surgery because of bleeding, perforation or other complications. In all, about 95% of all cases can be performed laparoscopically, while 5% of laparoscopic cases can result in a conversion to the open procedure.
1-Bleeding.
2-Perforation of oesophagus.
3-Perforation of stomach.
4-Splenic injury.
These complication is rare and account less than 2%.Complications more in open procedure than that of laparoscopic procedure.
After Surgery :
When patients wake up from surgery, they often have a NG tube to decompress the area around the wrap. Their diet is restricted to liquids for the first day after the tube is removed. A soft diet is recommended for several weeks after surgery.
Hospital Stay and Recovery :
Most patients are able to return home the first or second day after laparoscopic surgery and 4 to 7 days after the open operation. Return to full activity usually takes 1 to 2 weeks following most laparoscopic anti-reflux repairs and 4 to 6 weeks after an open repair. For about 2 weeks after surgery, patient will need to take an acid reducing medication such as Zantac. A follow up appointment should be made with the surgeon 7 to 10 days after discharge so that your questions can be answered, and progress can be assessed and patient can be examined.
Since the operation creates a valve mechanism at the bottom of the esophagus to prevent reflux, it may also cause resistance to the passage of food causing more air to be swallowed than before surgery. Therefore, following fundoplication, patients often experience periods of gas-bloat syndrome. During these episodes, which can last up to 2 to 3 hours, an increase in swallowed air makes it difficult to belch or vomit. In addition, patients often experience abdominal distention, nausea and an increase in flatulence. The soft diet should help prevent this syndrome.
In addition, for about 6 weeks after the laparoscopic repair, patients may experience dysphagia (difficulty swallowing) due a post-surgical swelling at the wrapped site.
Although dysphagia is almost always temporary, 2% of patients experience long term symptoms. These patients may be treated with balloon stretching of the area. Discusion :
For surgical management of GERD regardless of the way of access the main and popular procedure, that is wrapped of fundus around the oesophegus, is same in both laparoscopic and open surgery[1]. The operating time is significantly shorter for laparoscopy group in comparison with the open repair group (99 versus 142 minutes)[2-4]. Estimated blood loss is more in open surgeries (120 vs 95 ml)[6]. Rate of conversion to open surgery is about 5% [6] and the most common reasons are due to failure to progress or due to the complication that occur during operation(oesophgeal perforation,gastric perforation,splenic injury)[12] . Conversions happened more with surgeons with less experience in successful laparoscopic repair. complicaton,convertion rate decrease with increase experience of surgeon[1]. In those patients who underwent laparoscopy, postoperative comfort is obviously more and the amount of narcotic used is significantly lesser than open group.[2] Laparoscopic group have lower wound infection rate[13] Length of hospital stay is obviously shorter in laparoscopic group (3 vs 7 days) and they returned to their work earlier(14 vs 30 days)[15]. Cosmetically, scars following laparoscopic surgery are much more acceptable and patients are more satisfied with their operation [18] . Temperory dysphagia is more in laprascopic procedure,but long term dysphagia is the same[10]
Conclusion :
GERD is an extremely common disease that affects millions of people. Most patients can control their symptoms of heartburn with lifestyle changes and medications. The major drawback to medical therapy is its inability to address the underlying problem of a structurally defective LES. At this time, only surgery can improve the function of the LES.
Laparoscopic fundoplication is an option that more patients and their physicians are accepting as an alternative to a lifetime of medication. It is effective therapy for patients with GERD, and for some patients, may be more effective than medical therapy at controlling their symptoms and allow them to resume a normal lifestyle. Laparoscopic antireflux surgery should be strongly considered in patients with poorly controlled reflux, young patients, those with complications from their reflux, and those with atypical reflux symptoms.
From a functional point of view, both techniques were equally effective except concerning belching ability and temporary dysphagia .
References :
1. Watson DI, Jamieson GG, DeVitt PG, et al. Changing strategies in the performance of laparoscopic Nissen fundoplication as a result of experience with 230 operations. Surg Endosc. 1995;9:961-966.
2. Kauer WK, Peters JH, DeMeester TR, Heimbucher J, Irel and AP, Bremner CG. A tailored approach to antireflux surgery . J Thorac Cardiovasc Surg. 1995;110:141-147
3. Spechler SJ. Comparison of medical and surgical therapy for complicated gastroesophageal reflux disease in veterans. N Engl J Med. 1992;326:786-792
Rattner DW, Brooks DC. Patient satisfaction following laparoscopic and open antireflux surgery . Arch Surg. 1995;130:289-294
4. DeMeester TR, Bonavina L, Albertucci M. Nissen fundoplication for gastroesophageal reflux disease: evaluation of primary repair in 100 consecutive patients. Ann Surg. 1986;204:9-20.
5. Frantzides CT, Carlson MA. Laparoscopic versus conventional fundoplication. J Laparosc Surg. 1995;3:137-143
6. Luostarinen M, Isolauri J, Laitinen J, et al. Fate of Nissen fundoplication after 20 years: a clinical, endoscopical, and functional analysis. Gut. 1993;34:1015-
1020.7. Richards KF, Fisher KS, Flores JH, Christensen BJ. Laparoscopic Nissen fundoplication: cost, morbidity, and outcome compared with open surgery . Surg Laparosc Endosc. 1996;6:140-143.
7. Richards KF, Fisher KS, Flores JH, Christensen BJ. Laparoscopic Nissen fundoplication: cost, morbidity, and outcome compared with open surgery . Surg Laparosc Endosc. 1996;6:140-143.
8. Hunter JG, Trus TL, Branum GD, Waring JP, Wood WC. A physiologic approach to laparoscopic fundoplication for gastroesophageal reflux disease. Ann Surg. 1996;223:673-687
9.DeMeester TR, Johnson LF, Joseph GJ, Toscano MS, Hall AW, Skinner DB. Patterns of gastroesophageal reflux in health and disease. Ann Surg. 1976;184:459-470
10. Salminen JT, Salo JA, Tuominen J, Rämö OJ, Färkkilä M, Mattila S. pH-metric analysis after successful antireflux surgery : comparison of 24-hour pH profiles in patients undergoing floppy fundoplication or Roux-en-Y duodenal diversion. J Gastrointest Surg. 1997;1:494-498
12. Peters JH, Heimbucher J, Kauer WK, Incarbone R, Bremner CG, DeMeester TR. Clinical and physiologic comparison of laparoscopic and open Nissen fundoplication (see "Comments"). J Am Coll Surg. 1995;180:385-393
13.omparison of Long-term Outcome of Laparoscopic and Conventional Nissen Fundoplication. Annals of Surgery 246(2)
14.Omura, Nobuo (2007) Gastric Ulcer After Laparoscopic Fundoplication for Gastroesophageal Reflux Disease: Significance of the Eradication of Helicobacter pylori. Surgical Laparoscopy Endoscopy & Percutaneous Techniques 17(3)
15.Violette, A. (2007) Quality of life convergence of laparoscopic and open anti-reflux surgery for gastroesophageal reflux disease. Diseases of the Esophagus 0(0)
16.Omura, N. (2007) Prediction of recurrence after laparoscopic fundoplication for erosive reflux esophagitis based on anatomy-function-pathology (AFP) classification. Surgical Endoscopy 21(3)
17.Nissen R: Eine ainfache operation zur beeinflussung der refluxoesophagitis. Schweiz Med Wochenschr. 1956;86-:590-2.
18.Dallemagne B, Weerts JM, Jehaes C, et al. Laparoscopic Nissen fundoplication: Preliminary report. Surg Laparosc Endosc 1991;1(3):138-43.
19.Cuschieri A, Hunter J, Wolfe B, et al. Multicentre prospective evaluation of laparoscopic antireflux surgery: Preliminary report. Surg Endosc 1993;7:505-10.
20.Coller D, Cadiere GB. Conversions and complications of laparoscopic treatment of gastroesophageal reflux disease. Am J Surg 1995;169:622-6.
21.Dallamagne B, WeertsJM, Jeahaes C, et al. Results of laparoscopic Nissen fundoplication. Hepatogastroenterology 1998;45:1338-43.
22.Edye M, Salky B, Posner A, et al. Sac excision is essential to adequate laparoscopic repair of paraoesophageal hernia. Surg Endosc 1998;12:1259-63.
23.Booth MI Jones L. Results of laparoscopic Nissen fundoplication at 2-8 years after surgery. Bri J of Surg 2002;89:476-81.
24.Monabe N. Hazurro K. The increasing incidence of reflux oesophagitis during the past 20 years in Japan . Gastroenterology 1999;166:A244.
25.Walsh JB, et al. Patient outcomes and dysphagia after laparoscopic anti-reflux surgery performed without use of intra-op esophageal dilators. Am Surg 2003;69(3):219-23.
26.O Boyle CJ Watson DI De Beaux AC. Pre-op predictors of long term outcome after lap. fundoplication. Aust NJ Surg 2002;72:471-5.
27.Khajanchi YS. Urbach DR. Butler . Laparoscopic anti-reflux surgery in the elderly. Surgical outcome and effect on quality of life. Surg Endosc 2002;16:25-30.
28.Hent J Brun J Fendrich AM. An evidence based appraisal of reflux disease management. The Genval workshop report. Gut 1999;Suppl 2;44.






