Dr. Hanom Husni Syam
Obstetrician and Gynecologist Specialist
Diploma in Minimal Access Surgery (Laparoscopy)
Member World Association of Laparoscopic Surgeon (WALS)
Department Obstetric and Gynecology Trisakti University Jakarta
Teratai Fertility (IVF) Clinic Gading Pluit Hospital Jakarta
Abstract
BACKGROUND: The laparoscopic approach to myomectomy has raised questions about the risk of uterine rupture in patients who become pregnant following surgery. It has been suggested that the rupture outside labour in pregnancies following laparoscopic myomectomy can be due to the difficulty of suturing or to the presence of a haematoma or to the wide use of radio frequencies.
AIM: To assess the outcome of pregnancy following laparoscopic myomectomy
METHODS: A literature search performed using engine Google, High wire press, Springer link, and Yahoo. Selected papers screened for other related reports.
RESULTS: There were no incidents of uterine scar rupture in any of these studies.
CONCLUSIONS: Uterine rupture during pregnancies following laparoscopic myomectomy is rare. This review article did not confirm the hypothesis that laparoscopic myomectomy is associated with an increased risk for uterine dehiscence during pregnancy.
KEYWORDS: laparoscopic myomectomy, pregnancy, and uterine rupture
INTRODUCTION
Nowadays, laparoscopic myomectomy has become the elective procedure in selected patients. Laparoscopy effectively shortens the hospital stay and avoids the major risk of the classical route, i.e. adhesion formation. Laparoscopic myomectomy (LM) is a recently introduced technique that enables intramural and subserous myomas <9 cm in size and few in number to be managed by surgery. The rate of complications in the short term is low, provided that the surgeons are suitably trained (Dubuisson et al., 1996). Compared with myomectomy by laparotomy, LM offers reduced postoperative pain, a shorter hospital stay, and quicker return to normal activity (Mais et al., 1996). When pregnancy is desired, the technique appears particularly advantageous in that it could reduce the risk of postoperative adhesions compared with laparotomy (Bulletti et al., 1996). In selected cases, laparoscopic myomectomy has been reported to be an effective technique that is associated with a low rate of patient morbidity (Dubuisson et al., 1996). Because myomectomy is often performed to preserve the uterus for future pregnancy, maintaining the integrity of the uterine wall is of utmost importance (Dubuisson et al., 1995).
It is found that there is an increasing concern over the incidence of uterine rupture in pregnant women with a history of an earlier laparoscopic myomectomy. The fact that uterine rupture has been reported remote from term and following myomectomies performed for subserous and even pedunculated myomas (Dubuisson et al., 2000) are especially worrying. Uterine rupture has also been reported to occur without signs of fetal distress. Most cases of uterine rupture have been described as isolated case reports, and several case series have had no or very low rates of this complication (Dubuisson et al., 2000; Seinera et al., 2000).
OPERATIVE TECHNIQUES: >The difficulties in the operation, as with myomectomy by laparotomy, are the risk of per-operative haemorrhage and the prevention of post-operative adhesions. Use of the laparoscopic route for the myomectomy also raises certain particular problems connected with this approach: bloodless enucleation of the myomata is absolutely essential and a perfect suture must be achieved to obtain a good quality scar. There are several principles to use of the LM technique (Dubuisson et al., 2000)
- The principles of microsurgery must be applied to LM: avoidance of intraperitoneal contamination; use of fine and atraumatic instruments; gentle and atraumatic manipulation of the uterus without grasping the pelvic organs (except the myoma itself).
- When performed LM, each myoma must be excised via its own hysterotomy: it cannot use the same technique as myomectomy by laparotomy that is, removing all the myomata present on the uterus via an anterior sagittal hysterotomy.
- Dissection must take place in every case along the cleavage plane separating the myoma from the adjacent myometrium. This cleavage plane is bounded by a pseudo-capsule made up of compressed muscular fibres and diverted uterine vessels. This allows healthy adjacent myometrium to be preserved and damage avoided to the peri-myomatous vessels which are often distended due to compression by the myoma and could be the origin of considerable haemorrhage.
- Electrocoagulation must be used as sparingly as possible to achieve haemostasis of the edges after myomectomy. Certain cases of uterine rupture during pregnancy reported after LM and after myolysis suggest that the use of electrocoagulation may induce necrosis of the myometrium resulting in a post-operative fistula.
Suture of the hysterotomy must always respect a certain number of principles. Indeed any technical deficiency when carrying it out may result in uterine rupture during a subsequent pregnancy. Apart from pedunculated myomata, the myomectomy sites must always be sutured. In the experience of certain teams at the beginning, when no suture was carried out, the resulting scars were fine or dehiscent. The uterine suture does not necessarily have to use several planes, despite the recommendation of certain authors. The suture must always take up the full depth of the edges of the hysterotomy and result in total contact over the whole of the myomectomy defect in order to avoid secondary constitution of a haematoma deep inside the myometrium. This kind of haematoma can cause weakness in the scar tissues and the constitution of a secondary fistula. When the uterine cavity has been opened or when the myomectomy defect is deep, it is necessary to make a suture in two planes. It is possible to make this type of suture in several planes by laparoscopy. However, if this proves difficult there should be no hesitation in using laparoscopic assisted myomectomy (LAM) to complete it successfully. This procedure is an intermediate procedure between laparotomy and LM: laparoscopy is used to help myoma(ta) exposure; to begin or achieve enucleation; the uterine suture is then carried out by mini-laparotomy in a traditional fashion.
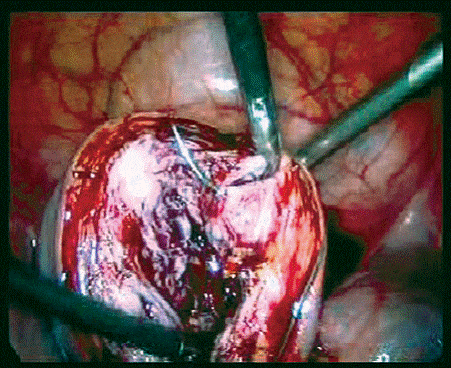
Figure 1. Suturing the cut edges after myomectomy.
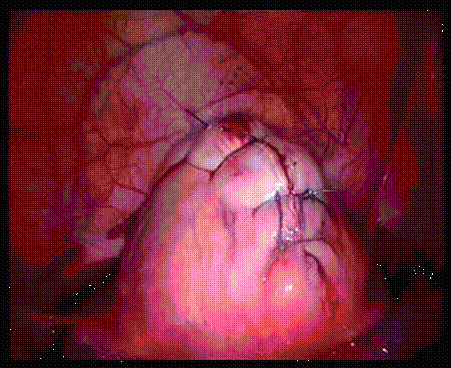
Figure 2. After closure of myometrium.
Myomectomi was performed with a standard technique using three suprapubic ports. The uterus was always cannulated to allow the correct exposure of myomas. For pedunculated myomas, the pedicle was secured using a pre-tied or extracorporeally-tied loop and coagulated and transected with bipolar forceps and scissors. To decrease vascularization and blood loss, starting in 1997 Rossetti et al. injected myomas with diluted (1: 100) ornithine vasopressin. For subserous and intramural myomas, they carried out the serosal incision vertically over the convex surface of the myoma using a monopolar hook. After exposure of the myoma pseudocapsule, grasping forceps were positioned to apply traction to the myorna and expose the cleavage plane. Enucleation was carried out by traction on the fibroid and by division with a unipolar hook or mechanical cleavage. Hemostasis during dissection was achieved by bipolar coagulation. Suturing was usually done along one or two layers depending on the depth of incision with interrupted, simple or more frequently cross-stitches tied intracorporeally using 1 or 0 Polyglactin sutures.
Material and Methods
A literature search was performed using Highwire press, Pubmed, the search engine Google and Online Springer facility available at World Laparoscopy Hospital, Gurugram, NCR Delhi. The following search terms were used: “Laparoscopic myomectomy”, “Pregnancy”, “Uterine rupture” and “Pregnancy outcomes”. Selected papers were screened for further references. Criteria for selection of literature were the number of cases (excluded if less than 20), methods of analysis statistical or non-statistical, operative procedure only universally accepted procedures were selected and the Institution where the study was done (Specialized institution for laparoscopic myomectomy were given more preference)
Results
Kucera E et al. (2006) in their report analyzed 69 patients after LM. The conception rate after LM was 56.5%. They didn't observe any increased incidence of fetomaternal morbidity or severe pregnancy and labour related complications. There was no uterine rupture after LM in their group. The cesarean section was rate 44.8%. LM in infertile patient is one of the most common surgical procedures. The appropriate surgical management of uterine scar is mandatory. Skilled reproductive surgeon must perform this operation. The pregnancy following LM is at high risk with increased caesarean section rate.
Paul PG, et al. (2006) reported that uterine rupture during pregnancies following laparoscopic myomectomy is rare following single-layer myometrial closure. Of the 217 women followed up, 115 had pregnancies subsequent to a laparoscopic myomectomy. Of 141 pregnancies, there were 87 Caesarean sections, 19 vaginal deliveries, 29 abortions and 6 ectopic pregnancies. There were no incidents of uterine scar rupture in any of these pregnancies.
Goldberg J et al. (2006) showed that although most pregnancies following uterine artery embolization have good outcomes, myomectomy should be recommended as the treatment of choice over uterine artery embolization in most patients desiring future fertility. Pregnancy rates following myomectomy, both via laparoscopy and laparotomy, are in the 50-60% range, with most having good outcomes. Both myomectomy and uterine artery embolization are safe and effective fibroid treatments, which should be discussed with appropriate candidates. Pregnancy complications, most importantly preterm delivery, spontaneous abortion, abnormal placentation and postpartum hemorrhage, are increased following uterine artery embolization compared to myomectomy.
Seracchioli R et al. (2006) reported that of the 514 women followed up, 158 pregnancies were achieved. There were 43 (27.2%) spontaneous abortions, 4 (2.6%) ectopic pregnancies, and 1 (0.6%) therapeutic abortion. Only 27 patients (25.5%) had vaginal deliveries, whereas 79 (74.5%) underwent cesarean section. No instances of uterine rupture were recorded. Their preliminary results confirmed that LM, performed by an expert surgeon, could restore reproductive capacity, allowing patients to have a successful pregnancy.
Campo S et al. (2003) analyzed that myomectomy significantly improves pregnancy outcome in patients with subserous or intramural fibroids, probably removing a plausible cause of altered uterine contractility or blood supply. Out of 128 patients submitted to myomectomy, we considered eligible for this study only the 41 patients wishing to conceive after surgery and who did not present any plausible infertility factor, apart from the removed myomas. Their results suggest that the main determinants of pregnancy rate after surgery are patient age, diameter and intramural localization of the myomas and type of surgery.
Soriano D et al. (2003) found that of 106 infertile women with uterine leiomyomas, of whom 88 women underwent laparoscopic myomectomy and 18 laparoconversion. No difference in the pregnancy rate was noted between the laparoscopic and laparoconversion groups (48 and 56%, respectively). There was no difference between the two groups as regards the rates of pregnancy-related complications and vaginal delivery. No uterine rupture occurred. They concluded that laparoscopic myomectomy is feasible and safe, and should be considered for infertile women with uterine fibroids. Fertility and pregnancy outcomes following laparoscopic myomectomy are comparable with those following myomectomy after laparoconversion.
Landi S et al. (2003) described that of 72 women were pregnant at least once after laparoscopic myomectomy. Four women conceived twice and four are pregnant as of this writing. One multiple pregnancy occurred. Twelve pregnancies resulted in first-trimester miscarriage, one in an ectopic pregnancy, one in a blighted ovum, and one in a hydatiform mole. One patient underwent elective first-trimester termination of pregnancy. Thirty-one women had vaginal delivery at term and 26 were delivered by cesarean section. No case of uterine rupture or dehiscence occurred.
Stringer NH et al. (2001) found that Laparoscopic suturing of the endometrial cavity in three layers does not prevent future pregnancies, and pregnancies can progress to term and in some cases be delivered vaginally without dehiscence.
Dubuisson JB et al. (2000) found that ninety-eight patients became pregnant at least once after LM, giving a total of 145 pregnancies. Among the 100 patients who had delivery, there were three cases of spontaneous uterine rupture. Because only one of these uterine ruptures occurred on the LM scar, the risk of uterine rupture was 1.0% (95% CI 0.0-5. 5%). Seventy-two patients (72.0%) had trials of labour. Of these, 58 (80.6%) were delivered vaginally. There was no uterine rupture during the trials of labour. Spontaneous uterine rupture seems to be rare after LM. When performing LM, particular care must be given to the uterine closure.
Seinera et al. (2000) described that the pregnancy outcome of 54 patients submitted to laparoscopic myomectomy at their Institution and prospectively followed during subsequent pregnancies. A total of 202 patients underwent laparoscopic myomectomy. A total of 65 pregnancies occurred in 54 patients who became pregnant following surgery. No cases of uterine rupture occurred. A Caesarean section was performed in 45 cases. In terms of the safety of laparoscopic myomectomy in patients who become pregnant following surgery, their results were encouraging. They suggested that further studies are needed to provide reliable data on the risk factors and the true incidence of uterine rupture.
Nezhat CH et al. (1999) analyzed that of the 115 women, there were 42 pregnancies in 31 patients. Two women were lost to follow-up. Of the remaining 40 pregnancies, six ended with vaginal delivery at term. Caesareans were performed in 22 cases, including 21 at term and one at 26 weeks gestation. Two pregnancies were associated with a normal delivery, but the mode of delivery is unknown. Eight resulted in first trimester pregnancy loss, one was an ectopic pregnancy, and one patient underwent elective termination. Spontaneous uterine rupture was not noted during pregnancy or at term in any of the cases. Our series did not confirm the hypothesis that laparoscopic myomectomy is associated with an increased risk for uterine dehiscence during pregnancy.
Dubuisson JB et al. (1996) reported that the overall rate of intrauterine pregnancy, after laparoscopic myomectomy, was 33.3% (seven patients). Out of the seven pregnancies, four were spontaneous and began within 1 year of the operation. The other three were achieved after in-vitro fertilization in patients with associated infertility factors. In the four patients who gave birth by Caesarean section, no adhesions were found on the myomectomy scar. From these preliminary results, laparoscopic surgery for myomas seems to offer comparable results with those obtained by laparotomy. No uterine rupture was observed.
Ribeiro SC et al. (1999) Laparoscopic myomectomy can be offered to patients who want to have children and who refuse to undergo an abdominal myomectomy. Patient selection as well as meticulous surgical technique is the key factors in achieving a successful outcome.
Daraï E et al. (1997) reported that of 19 pregnancies were obtained in 17 patients after laparoscopic myomectomy (38.6%): eight vaginal deliveries, three Caesarean sections, four miscarriages, two abortions, one ectopic pregnancy and one therapeutic abortion. No uterine rupture was noted. Pelvic adhesions were found in the four patients who underwent second-look procedure. Their preliminary results indicate that laparoscopic myomectomy is a useful technique.
Discussion
Myomectomy is a challenging procedure because it involves the reconstruction of an organ that can undergo remarkable structural changes, as it does in pregnancy. The literature documents normal reproductive performance of uteri after laparotomic myomectomy (Li et al., 1999). Paul et al. (2006) found that the frequencies of early pregnancy losses and preterm deliveries in their series were within normal limits, though that for ectopic pregnancies was higher (4.3%). This is consistent with the higher incidence of ectopic pregnancies in patients with infertility (Pisarska and Carson, 1999). Nezhat et al. (1999) found that in their series, the observed frequency of miscarriages, ectopic pregnancies and preterm deliveries was within normal limits. The present 19% miscarriage rate matches the 19% reported after myomectomy at laparotomy (Buttram and Reiter, 1981).
Most studies have reported an increased incidence of Caesarean section (Hurst et al., 2005). This is not unexpected in the presence of a scarred uterus. In addition, most patients have a history of infertility and are in the older age groups. This though does not make myomectomy a mandatory indication for elective Caesarean sections, high vaginal delivery rates have been achieved in studies by Dubuisson et al., (2000). Recommendations for a waiting period before attempting pregnancy to ensure adequate wound healing though recommended have been questioned (Landi et al., 2003) and are not backed by good evidence. Paul et al. 2006 showed that the majority of their patients conceived in the first year after surgery (82.6%) and a significant number in the first six months (55.6%). Nezhat et al. 1999 described that the increased incidence of Caesareans is not surprising, since this is the recommended method of delivery for women in whom the uterine wall has been deeply penetrated. All of the patients who delivered vaginally had pedunculated or subserosal myomas.
Pregnancies following any surgical procedure involving the uterus have an increased risk of rupture or dehiscence during pregnancy and labour. Such risks in relation to Caesarean sections have been well quantified. This has helped in improved management of post-Caesarean pregnancies before and during labour. The same cannot be applied in cases of women with a previous history of myomectomy, whether open or laparoscopic, because of the absence of good quality studies. One possible cause of uterine rupture after laparoscopic myomectomy is the wide use of electrosurgery that may result in poor vascularization and tissue necrosis with an adverse effect on scar strength (Nezhat et al., 1996). Electrosurgery was used to remove the myoma and obtain haemostasis in five out of the six reported uterine ruptures. In one case the uterus ruptured at 26 weeks following laparoscopic myolysis of a 3 cm intramural myoma (Arcangeli and Pasquarette, 1997). Myolysis is an endoscopic technique in which the tumour is coagulated with the help of bipolar probes inserted into the myoma. In the reported case there was no suture of the uterine wound.
Although many studies did not show any cases of uterine rupture, the occurrences mentioned above should serve as a warning. Considering that the procedure of laparoscopic myomectomy is rather new, it may not be efficacious for patients who desire future pregnancy, especially when performed by the novice endoscopic surgeon. In any case, laparoscopic myomectomy should be performed cautiously. Excess thermal damage should be avoided and adequate uterine repair must be assured using multiple layer suturing techniques. Both thermal damage and haematoma formation have been blamed as causes for sub-optimal healing and rupture during a future pregnancy. (Dubuisson et al., 2000; Landi et al., 2003). Thermal damage has been especially blamed in cases where subserous myomas were removed (Nkemayim et al., 2000). Correct reapproximation is not dependent on the number of layers of sutures but on the technique of full thickness, evenly spaced suture placement, thus avoiding haematoma formation. Overaggressive control of bleeders using electrosurgical techniques should be avoided. Increasing the numbers or layers of sutures without adhering to the above principles may in fact compromise healing by causing tissue ischaemia. Paul et al., 2006, found that uterine rupture following laparoscopic myomectomy is rare following single-layer myometrial closure. This data though reassuring are not conclusive, and there is a need for a randomized study to compare single-layer and multi-layer suturing techniques.
Conclusions
LM enables surgical treatment of subserous and intramural myomata of average size (<9 cm) and few in number. When the surgeons are sufficiently experienced this technique does not involve a greater risk of pen-operative complications. LM might reduce the risk of post-operative adhesions compared with laparotomy, which is a considerable advantage in a context of surgery for infertility. At present there is not enough evidence to say whether the hysterotomy scars after LM are as strong as those obtained after laparotomy. The risk of uterine rupture after LM seems low, however, and should not constitute a counter-indication for this operation if pregnancy is desired, provided that the myomectomy is truly justified. If good quality uterine scar is to be obtained, the surgeons need to be very experienced in laparoscopic surgery and to pay particular care when suturing the uterus. Obstetricians dealing with antenatal patients with a history of a myomectomy before should be aware of possible complications and should consider management of such patients as in cases of post-Caesarean pregnancies.
References
- Arcangeli, S. and Pasquarette, M.M. (1997) Gravid uterine rupture after myolysis. Obstet. GynecoL, 89, 857.
- Bulletti, C, Polli, V, Negrini, V. et al. (1996) Adhesion formation after laparoscopic myomectorny. J. Am. Assoc. Gynecol. Laparosc, 3, 533-536.
- Buttram, V.C. Jr and Reiter, R.C. (1981) Uterine leiomyomata: etiology, symptomatology and management. Fertil. Steril, 36, 433–445.
- Campo S, Campo V, Gambadauro P. (2003) Reproductive outcome before and after laparoscopic or abdominal myomectomy for subserous or intramural myomas. Eur J Obstet Gynecol Reprod Biol. 110(2), 215-9.
- Daraï E, Dechaud, H, Benifla, J.L. et al. (1997) Fertility after laparoscopic myomectomy: preliminary results. Hum. Reprod, 12, 1931 1934.
- Dubuisson J, Fauconnier A, Deffarges J, Norgaard C, Kreiker G and Chapron C (2000) Pregnancy outcome and deliveries following laparoscopic myomectomy. Hum Reprod 15,869–873.
- Dubuisson, J.B, Chapron, C, Chavet, X. et al. (1996) Fertility after laparoscopic myomectomy of large intramural myomas: preliminary results. Hum. Reprod, 11, 518–522.
- Dubuisson, J.B, Chapron, C. and Levy, L. (1996) Difficulties and complications of laparoscopic myomectomy. J. Gynecol. Surg,. 12. 159-165.
- Dubuisson, J.B, Chavet, X, Chapron, C. et al. (1995) Uterine rupture during pregnancy after laparoscopic myomectomy. Hum. Reprod, 10, 1475-1477
- Goldberg J, Pereira L. (2006) Pregnancy outcomes following treatment for fibroids: uterine fibroid embolization versus laparoscopic myomectomy.Curr Opin Obstet Gynecol. 18(4), 402-6
- Hurst BS, Matthews ML and Marshburn PB (2005) Laparoscopic myomectomy for symptomatic uterine myomas. Fertil Steril 83,1–23.
- Kucera E, Dvorská M, Krepelka P, Herman H. Pregnancy after laparoscopic myomectomy--long-term follow up : Ceska Gynekol. 2006 Sep;71(5),389-93.
- Landi S, Fiaccavento A, Zaccoletti R, Barbieri F, Syed R and Minelli L (2003) Pregnancy outcomes and deliveries after laparoscopic myomectomy. J Am Assoc Gynecol Laparosc 10,177–181.
- Landi S, Fiaccavento A, Zaccoletti R, Barbieri F, Syed R, Minelli L. (2003) Pregnancy outcomes and deliveries after laparoscopic myomectomy. J Am Assoc Gynecol Laparosc. 10(2),177-81.
- Li, T.C, Mortimer, R. and Cooke, ID. (1999) Myomectomy: a retrospective study to examine reproductive performance before and after surgery. Hum. Reprod, 14, 1735 1740.
- Mais, V, Ajossa, S, Guerriero, S. et al. (1996) Laparoscopic versus abdominal myomectomy: a prospective, randomized trial to evaluate benefits in early outcome. Am. J. Obstet. Gynecot, 174, 654-658.
- Nehzat, F, Seidman, D.S, Nehzat, C. et at. (1996) Laparoscopic myomectomy today: why, when und for whom? Hum. Reprod, 11, 933 934.
- Nezhat, C.H, Nezhat, F, Roemisch, M. et at. (1999) Pregnancy following laparoscopic myomectomy: preliminary results. Hum. Reprod, 14, 1219 1221.
- Nkemayim DC, Hammadeh ME, Hippach M, Mink D and Schmidt W. (2000) Uterine rupture in pregnancy subsequent to previous laparoscopic electromyolysis: Case report and review of the literature. Arch Gynecol Obstet 264,154–156.
- Paul PG, Koshy AK, Thomas T. (2006) Pregnancy outcomes following laparoscopic myomectomy and single-layer myometrial closure. Hum Reprod., 21(12),3278-81
- Pisarska MD and Carson SA (1999) Incidence and risk factors for ectopic pregnancy. Clin Obstet Gynecol 42,2–8.
- Ribeiro, S.C, Reich, H, Rosenberg, J. et al. 1999) Laparoscopic myomectomy and pregnancy outcome in infertile patients. Fertil. Steril, 71, 571 574.
- Rossetti A, Sizzi O, Soranna L. (2001) Long-term results of laparoscopic myomectomy: recurrence rate in comparison with abdominal myomectomy. Hum Reprod, 16, 770—774
- Seinera P, Farina C and Todros T (2000) Laparoscopic myomectomy and subsequent pregnancy: results in 54 patients. Hum Reprod 15,1993–1996.
- Seracchioli R, Manuzzi L, Vianello F et al. (2006) Obstetric and delivery outcome of pregnancies achieved after laparoscopic myomectomy.Fertil Steril. 86(1),159-65.
- Soriano D, Dessolle L, Poncelet C, et al. (2003) Pregnancy outcome after laparoscopic and laparoconverted myomectomy. Eur J Obstet Gynecol Reprod Biol. 108(2),194-8.
- Stringer NH, Strassner HT, Lawson L, Oldham L, Estes C, et al. (2001) Pregnancy outcomes after laparoscopic myomectomy with ultrasonic energy and laparoscopic suturing of the endometrial cavity. J Am Assoc Gynecol Laparosc 8,129–136.






